

News and Events
аТЮХзЪбЖ
аТЮХзЪбЖ

2026-03-04
гХЕТ88 Immutailin? PVDFзЊгЁФЄаТЦЗЩЯЪа
дкЕААзжЪгыКЫЫсМьВтЗжЮіРћгУжаЃЌNC зЊгЁФЄЃЈЯѕЫсЯЫЮЌЫиФЄЃЉгыPVDF зЊгЁФЄЃЈОлЦЋЖўЗњввЯЉФЄЃЉЪЧСНЯюжїЬтЕФЙЬЯржЇГжНщжЪ
ЁЃЫќУЧЦОНшИїздЙжвьЕФЛњФмгХЪЦЃЌПэЗКРћгУгкПЦбаЫїЧѓгывНбЇеяЖЯСьгђЃЌЮЊЗжЦчЕФГЂЪджИБъЁ
ВщПДЯъЧщ
ВщПДЯъЧщ
242026.03
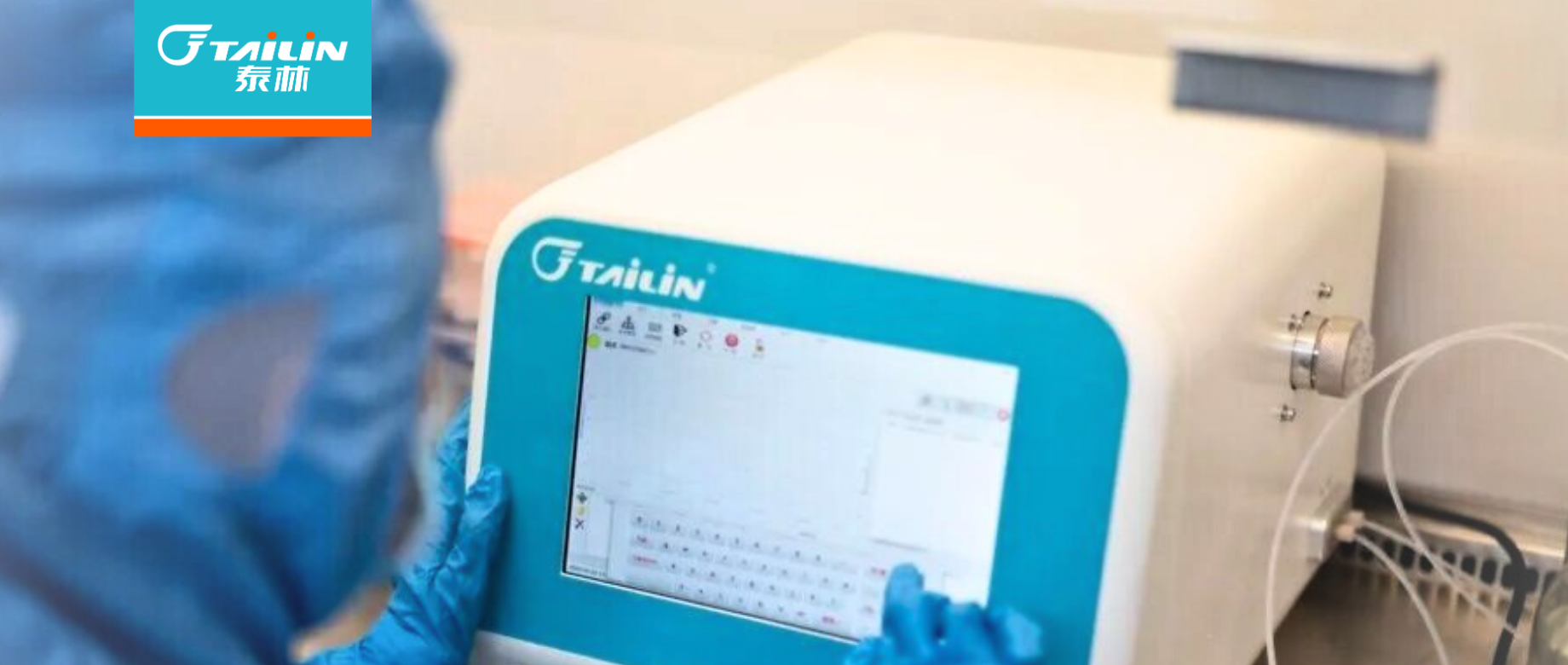
ШЈЭўШЯжЄ+жеЖЫШЯПЩЃЁгХЕТ88MD500ЮЂЩњЮяПьМьвЧЃЌгУЪ§ОнжўРЮдьвЉКЯЙцЗРЯп
НќШеЃЌеуНгХЕТ88ЗжЮівЧЦїгаЯоЙЋЫОзджїбаЗЂЕФMD500аЭЮЂЩњЮяМБОчМьВтЗжЮівЧЃЌВЛНіЫГРћЭЈЙ§еуНЪЁЪГЦЗвЉЦЗМьбщзъбадКЃЈвдЯТМђГЦЁАЪЁвЉМьдКЁБЃЉДњЬцВНжшбЇШЈЭўбщжЄЃЌИќНЋетЗнбщЁ
ВщПДЯъЧщ
132026.03
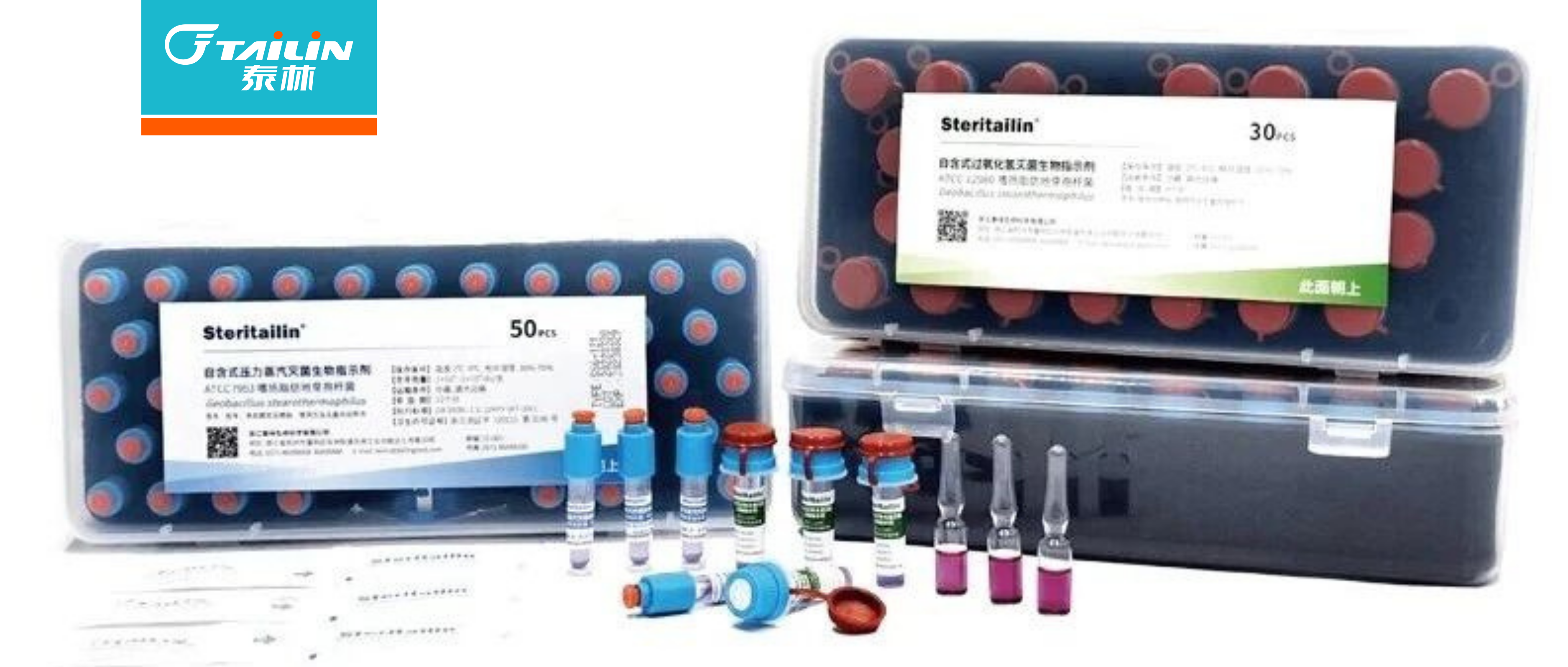
ЧГЮіЁЖGB/T 18281вНСЦБЃНЁВњЦЗУ№Оњ ЩњЮяХњЪОЮяЁЗЕФБфЖЏгыЕїећ
ЁЖGB/T 18281вНСЦБЃНЁВњЦЗУ№Оњ ЩњЮяХњЪОЮяЁЗЪЧгаЙиЩњЮяХњЪОМСЕФЭЈгУадКЭзЈгУадГпЖШ
ЁЃаТАцЁЖGB/T 18281-2024 вНСЦБЃНЁВњЦЗУ№Оњ ЩњЮяХњЪОЮяЁЗЯЕСаГпЖШгк2024Фъ11дТ28ШеЁ

ВщПДЯъЧщ
242026.02
ПЅЦєаТГЬЃЌТэСІШЋПЊЃЁ гХЕТ88ЩњЮяНёШеПЊЙЄРВЃЁ
ЗмНје§ЦфЪБЃЌПЊЙЄЦєаТГЬЃЁХЉРње§дТГѕАЫЩЯЮчЃЌгХЕТ88ЩњЮяаТДКПЊЙЄЕфРёдкИїИідАЧјЭЌВННјаа
ЁЃећИігХЕТ88ШЫДјзХаТДКЕФГЏЦјГСЗЕИкЮЛЃЌбИПьДгЁАМйЦкФЃЪНЁБЧаЛЛжСЁАЙЄзїФЃЪНЁБЃЌвдЛ№ШШЁ

ВщПДЯъЧщ
112026.02
2025гХЕТ88ФъЖШЗўЮёЛуБЈЁЊЁЊвдЗўЮёжЎЙтЃЌееССПЦММжЎТЗ
ЁАвдПЭЛЇЮЊжааФЁБЪЧгХЕТ88ЩњЮяЕФЗўЮёзкжМЮвУЧгХЛЏСїГЬЁЂДДаТЙцЛЎгУааЖЏкЙЪЭЗўЮёМлжЕгУзЈвЕгыЮТЖХзЎЕУПЭЛЇаХРЕетвЛФъРлМЦЗўЮё16,000+МвПЭЛЇЗўЮёзуМЃБщВМ10+ИіЙњЖШ30+ИіГЧЪа&nbЁ

ВщПДЯъЧщ
112026.02
?гХЕТ88ИКбЙИєРыЦїММЪѕдкИпЛюаддСЯвЉГіВњжаЕФРћгУ
вЛЁЂТЩР§ЖдЖОадвЉЦЗГіВњЕФгаЙиЛЎЖЈЁё WHO TRS-957 ANNEX 3 WHO good manufacturing practices for pharmaceutical products containing hazardous substanceЁ


 EN
EN
